EP204 Introduction to Material and Energy Balances Questions Answers
Hire EP204 Introduction to Material and Energy Balances Questions Answers Experts and Secure Grades
Question 1
Mass flow through a sonic nozzle is a function of gas pressure and temperature. For a given pressure p and temperature T, mass flow rate through the nozzle is given by the equation:
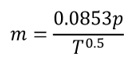
where m is in lbm/min, p is in atm, and T is in °R.
(a) Identify the unit for the constant ‘0.0853’.
(2 marks)
(b) Determine the new expression of the equation, if m, p and T are expressed in g/hr, mmHg and K, respectively.
(8 marks)
Question 2
Formic acid, CH2O2 (MW=46.025) is a colorless, clear, corrosive liquid with a pungent odor. It is widely used in used textiles and leather processing industries. For a given 75 wt % formic acid solution of specific gravity 1.86:
(a) Identify the mole fraction composition of this solution with a basis of 385 g solution.
(4 marks) (b) Estimate the volume (in gallons) of 3 mol CH2O 2 solution.
(6 marks)
Question 3
The heat capacity of liquid pentane is represented by the following correlation:
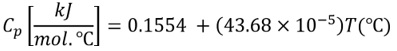
Express the correlation for Cp with the unit Btu/(lb-mol.°R) in terms of T in °R. [Hint: 1 lb-mole = 454 gmol]
(9 marks)
Question 4
A differential manometer filled with manometer fluid X is used to measure the pressure drop between two points in a process line containing liquid glycerol, C3H8O3 (SG=1.260). The pressure drop is found to be 13.5 mm Hg with the reading of the differential manometer is shown in Figure 4(a). In another occasion, a sealed end manometer (Figure 4(b)) containing manometer fluid X is used to measure the atmospheric pressure. Estimate h in cm, if the atmospheric pressure is 745 mm Hg.
(6 marks)
Question 5
Evaporation and recycling systems are adopted in the calcium chloride (CaCl 2) crystallization process. The simplified process flow diagram is depicted in Figure Q1.
The recycled stream (R) is maintained at the composition of 0.7 kg CaCl 2/kg H2O.
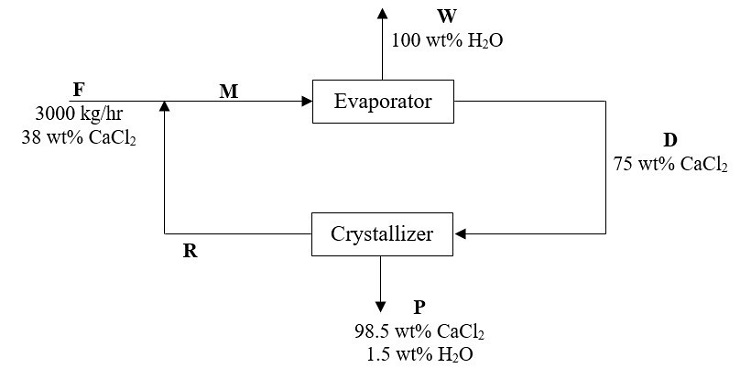
Figure Q1
(a) Sketch a completely labelled process flow diagram (with compositions) for the process.
(8 marks) (b) Compute the mass flow rate (kg/hr) of stream W, R and D, respectively.
(8 marks) (c) In the case of evaporator failure, it is found that the evaporator can only cope with the water evaporation rate (W) of 1.5 lbm/s. Estimate the new feed flow rate (F) required (lb m/s) and the formation rate (lbm/s) of crystal product (P), if the composition of all streams is to be remained unchanged.
(4 marks)
Question 6
A proposed process to remove H2S is by the reaction with SO 2 through the following equation:
2𝐻2𝑆(𝑔)+𝑆𝑂2(𝑔)→3𝑆(𝑠)+2𝐻2𝑂(𝑔)
In a test of the process, a gas stream containing 20 mole % H2S and 80 mole % CH4 is combined with a stream of pure SO 2. The process produces 3500 g of S (s) in an hour. In the product gas, the ratio of SO2 to H2S equals to 2 and the ratio of H2O to H2S is 5.
(a) Calculate the extent of reaction (mol/hr) of the process.
(3 marks)
(b) Determine the molar flow rates (mol/hr) for both feed gas streams and all components in the product gas stream by using extent of reaction method.
(12 marks) (c) Identify the limiting reactant.
(3 marks)
Question 7
A stream of 100 kg/min superheated steam enters a turbine at 20 bar and 450°C at a linear velocity of 80 m/s and leaves at a point 2 m below the turbine inlet at 5 bar and a velocity of 350 m/s. The heat loss from the turbine is estimated to be negligible, while the turbine develops 1800 kW of work. Determine the specific enthalpy, 𝐻̂ (kJ/kg) of the turbine outlet stream, if the 𝐻̂in of the inlet stream is 3358 kJ/kg.
(12 marks)
END OF QUESTION PAPER

